|
Unfortunately, the process for converting solar light into usable power is not perfect. PV modules are generally made by connecting several individual solar panels together to achieve useful levels of voltage and current, and putting them in a sturdy frame complete with positive and negative terminals. The final step is to install something that will protect the cell from the elements - often a glass cover plate. Silicon happens to be a very shiny material, which can send photons bouncing away before they've done their job, so an antireflective coating is applied to reduce those losses. There are a few more components left before we can really use our cell. With both current and voltage, we have power, which is the product of the two. The electron flow provides the current, and the cell's electric field causes a voltage. This causes further disruption of electrical neutrality, and if we provide an external current path, electrons will flow through the path to the P side to unite with holes that the electric field sent there, doing work for us along the way. If this happens close enough to the electric field, or if a free electron and free hole happen to wander into its range of influence, the field will send the electron to the N side and the hole to the P side. 
Each photon with enough energy will normally free exactly one electron, resulting in a free hole as well. When light, in the form of photons, hits our solar panel, its energy breaks apart electron-hole pairs. It's like a hill - electrons can easily go down the hill (to the N side), but can't climb it (to the P side). This electric field acts as a diode, allowing (and even pushing) electrons to flow from the P side to the N side, but not the other way around. Instead of having free electrons, P-type ("p" for positive) has free openings and carries the opposite (positive) charge. The other part of a typical solar cell is doped with the element boron, which has only three electrons in its outer shell instead of four, to become P-type silicon. N-type doped silicon is a much better conductor than pure silicon. The process of adding impurities on purpose is called doping, and when doped with phosphorus, the resulting silicon is called N-type ("n" for negative) because of the prevalence of free electrons. As a result, most of these electrons do break free, and we have a lot more free carriers than we would have in pure silicon. It takes a lot less energy to knock loose one of our "extra" phosphorus electrons because they aren't tied up in a bond with any neighboring atoms. However, there are so few of them in pure silicon that they aren't very useful.īut our impure silicon with phosphorus atoms mixed in is a different story. These electrons, called free carriers, then wander randomly around the crystalline lattice looking for another hole to fall into and carrying an electrical current. When energy is added to pure silicon, in the form of heat for example, it can cause a few electrons to break free of their bonds and leave their atoms. It doesn't form part of a bond, but there is a positive proton in the phosphorus nucleus holding it in place. It still bonds with its silicon neighbor atoms, but in a sense, the phosphorus has one electron that doesn't have anyone to hold hands with. Phosphorus has five electrons in its outer shell, not four. Consider silicon with an atom of phosphorus here and there, maybe one for every million silicon atoms. We usually think of impurities as something undesirable, but in this case, our cell wouldn't work without them. To address this issue, the silicon in a solar cell has impurities - other atoms purposefully mixed in with the silicon atoms - which changes the way things work a bit. In the process, you will learn why we're getting closer to using the sun's energy on a daily basis, and why we still have more research to do before the process becomes cost-effective. In this article, we will examine how solar panels generate electricity and exactly how solar panels work. If we could collect all of that energy, we could easily power our homes and offices for free and have reserves stored up with the excess energy produced. 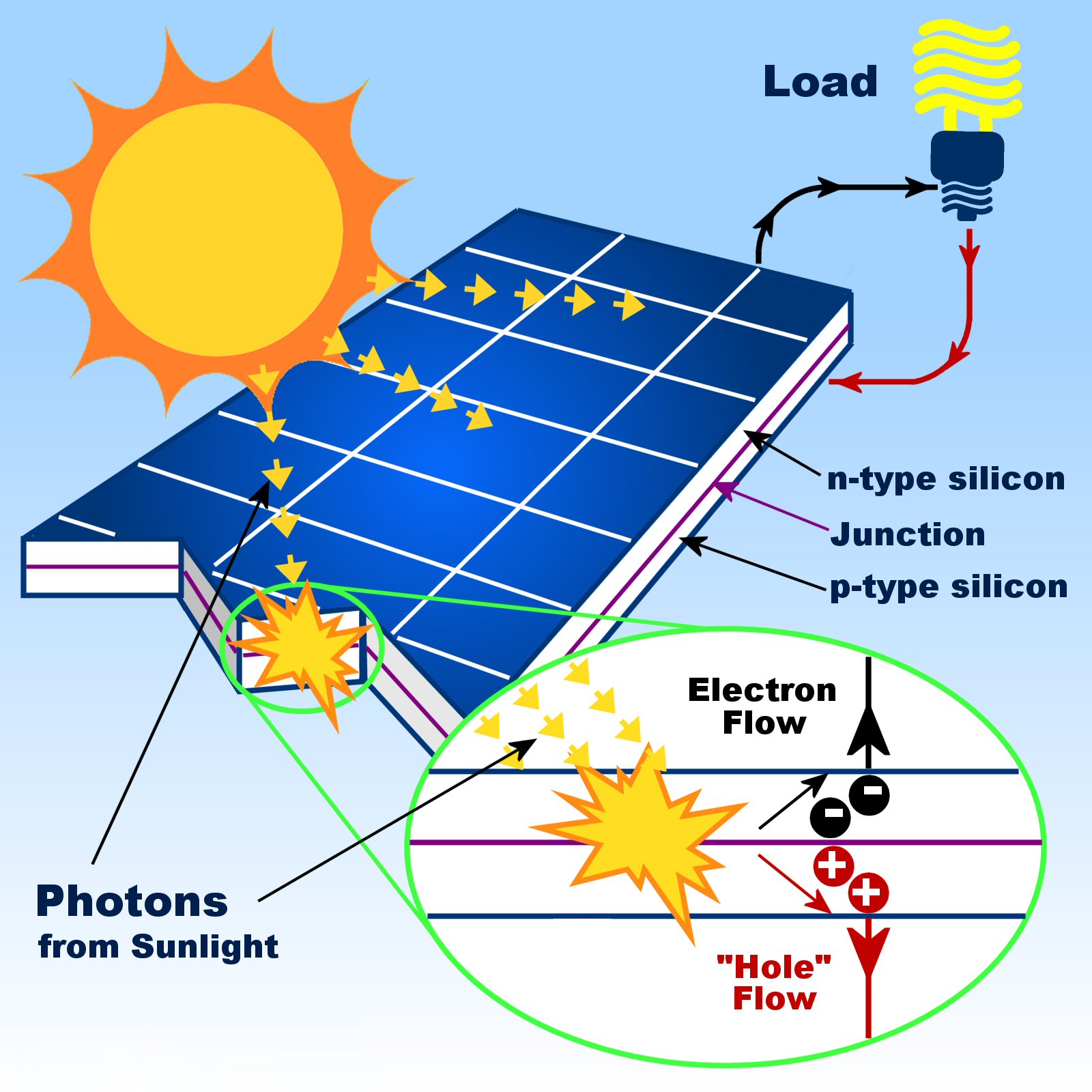
This is a seductive promise, because on a bright, sunny day,the sun's rays give off approximately 1,000 watts of energy per square meter of the planet's surface. The hope for a "solar revolution" has been floating around for decades - the idea that one day we'll all use free solar electricity from the sun. The technology continues to pop up in new devices all the time, from sunglasses to electric vehicle charging stations. They were once used almost exclusively in space, powering satellites' electrical systems as far back as 1958, but are being used more and more in less exotic ways.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
